A common mistake made in drawing organic compounds is exceeding the valency of a carbon atom:
Colloquially, carbon atoms with five covalent bonds are called Texas Carbons
Either because the Texas star has five points or because everything is bigger in Texas..
Formally, they are known as pentavalent carbons.
It turns out that well-formed pentavalent carbons are much more frequent than an organic chemistry class may have you believe.
SuperAcids
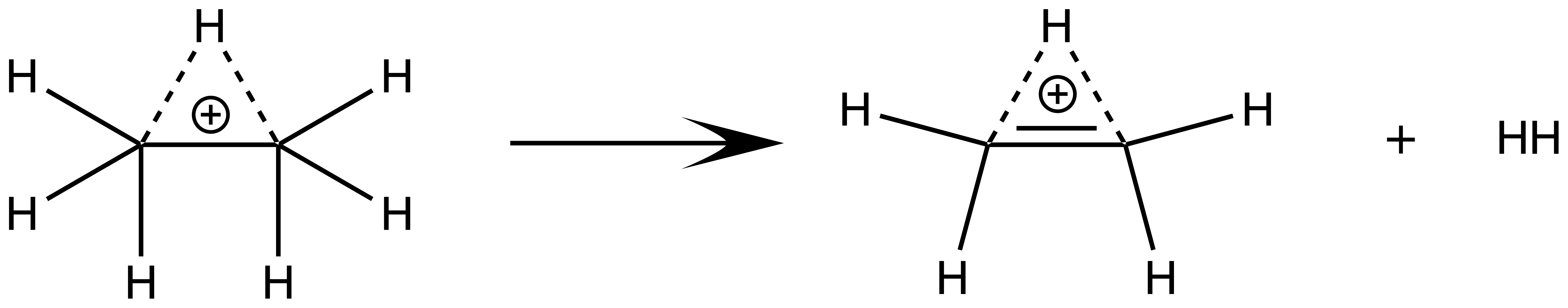
Superacidic cationic pentavalent carbons can be formed in the presence of very strong acids. The simplest pentavalent carbon, methanium (), can be prepared from methane in the presence of .
Ethanium () is a protonated ethane derivative, also hosting a pentavalent carbon. Ethanium is interesting partly because it is prepared using methanium:
and partly because it easily dissociates into ethenium, which also has pentavalent carbons:
In practice, pentavalent acids are formed as intermediate species and during thermal crackingPer a DOE report, page 8..
Both B and C
Yamamotoa's and Akibab's 2004 Synthesis of Hypervalent Pentavalent Carbon and Boron Compounds describes the characteristics of larger pentavalent compounds. It appears (somewhat expectedly) that bonds to well-charged, larger-size atoms like those in Group 15 and 16 help stabilize pentavalent carbon and boron.
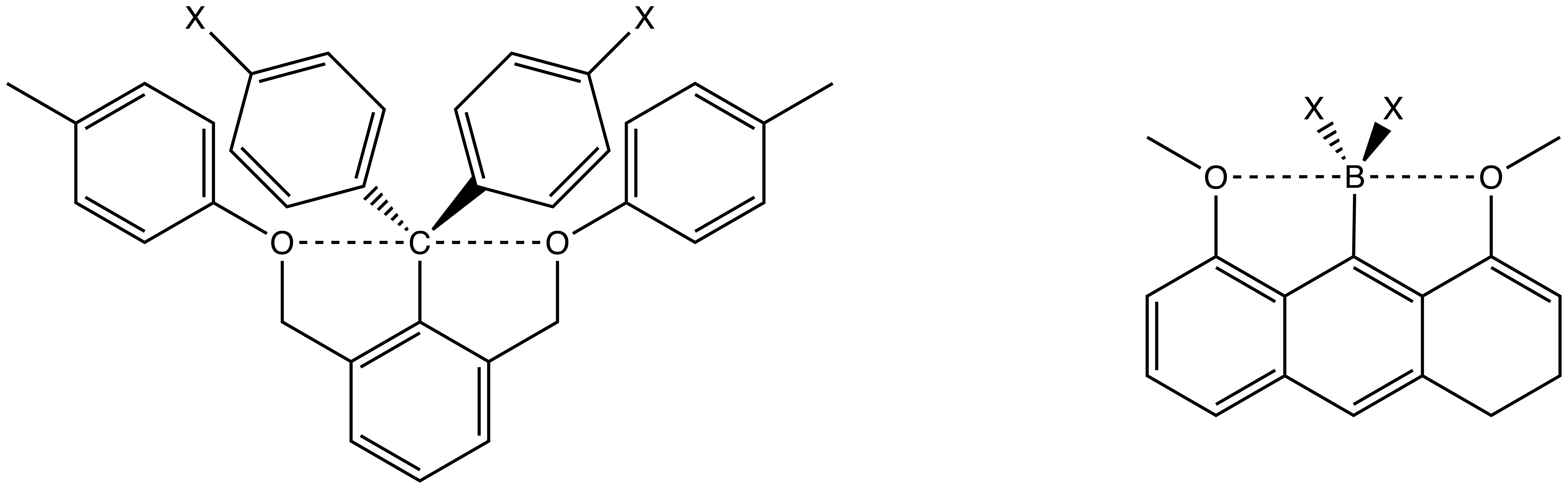
The bond angle of the emphasized is , more than the expected for a star-shaped pentavalency, and likely explained by the distance of the oxygens from the carbon.
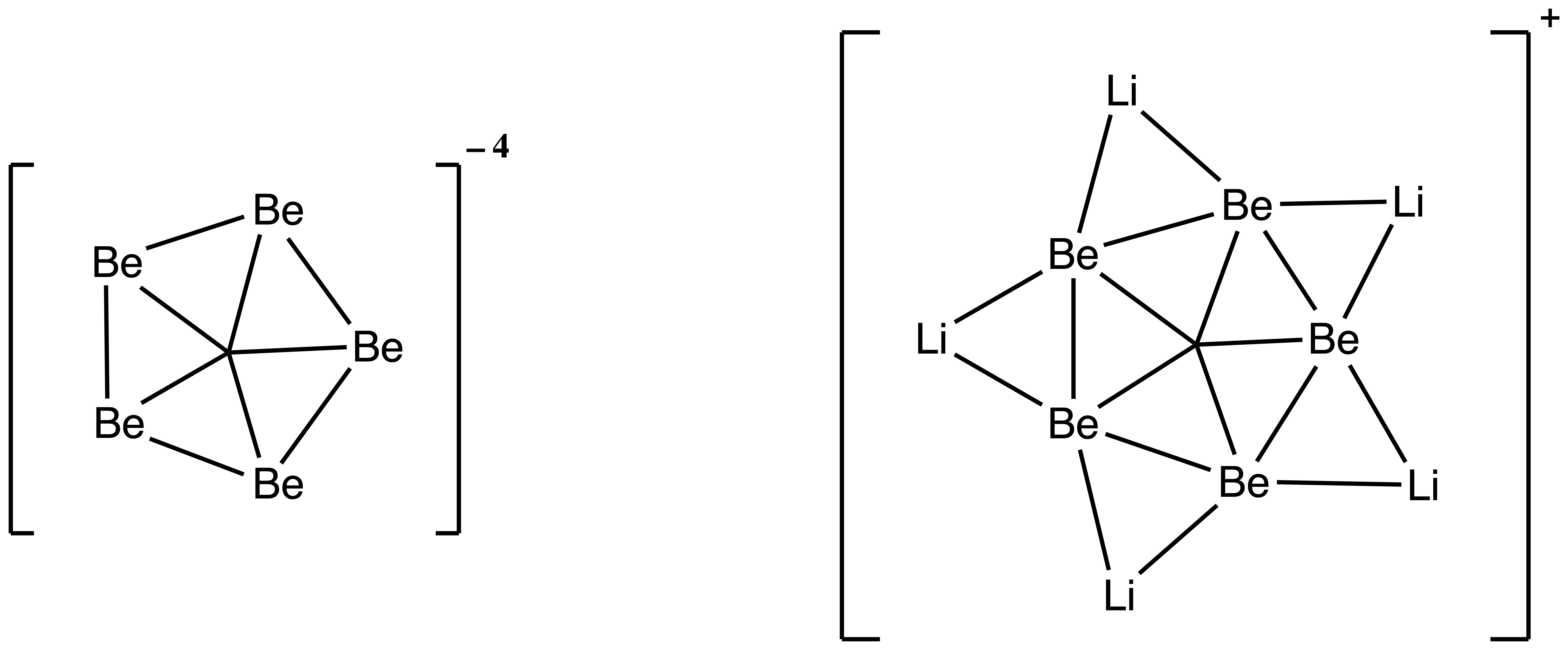
Are there star-shaped pentavalencies?
Yes! One is stable methanium, which we have already seen. Another is , which is predicted to be planar and possible to prepare experimentally in the gas phase. Grande-Aztatzi et al. predict the lithium cluster derivatives to also be planar. Both of these form beautiful five-point shapes.
Beyond the stars
Pentavalent carbons are one of those ideas most of us won't deal with very often. But I think unique patterns like these are worth knowing, at least for the sake of knowing.
If you're interested in hypervalencies, you may enjoy hexavalent carbons and undecavalent borons.